Sputnik over Europe
Since Sputnik V was registered in Russia in August 2020, a major global promotional campaign for the vaccine has been under way with the involvement of the state authorities, led by President Vladimir Putin. Recent weeks have seen the intensification of such a campaign in the European Union, which has been backed up by disinformation activities targeting Western vaccines. However, Sputnik V production levels have been low so far, and they do not meet the needs of either Russia or the countries that have signed up for it. Russian announcements that its production would soon begin outside the country, including in Europe, have also failed to come true. Available information shows that about a third of all Sputnik V doses delivered to the market are exported. However, an analysis of the contracts signed and the volume of deliveries made places Russia at the bottom of the global list of producers and suppliers. Like other manufacturers, it has to keep choosing who to send vaccines to first (and in what quantities). In effect, Europe and Latin America have been the priority destinations for shipments abroad, although even those contracts are not being fulfilled on time. Bulk quantities of Sputnik V are unlikely to arrive in Europe before autumn 2021.
The Russian promotional and disinformation offensive in Europe
Moscow is primarily striving to convince both publics and politicians that in the current situation, Sputnik V could be the solution to Europe’s problems with shortages of Western vaccines. The Russian authorities are promoting their own preparation at the bilateral level, offering member states either supplies of the vaccine itself or the technology to allow it to be manufactured in Europe. In recent months President Putin has discussed Sputnik V with the leaders of countries including Austria, France, Luxembourg, Germany and Serbia. Russian diplomacy is also trying to reach out directly to potential recipients of the jab, seeking support among foreign politicians or Russia-friendly institutions (such as the Russian-Italian Chamber of Commerce).
However, Moscow only began its efforts to obtain permission for Sputnik V to be marketed in the EU in early March, despite the fact that according to European Medicines Agency (EMA) procedures, it is possible to start the procedure for evaluating COVID-19 vaccines while they are still in trials. Moreover, unlike Western manufacturers who waited (or are still waiting) for the EMA to register their vaccines, Russia has managed to convince some countries to import Sputnik V under individual national authorisations.
At the same time, there are mounting reports that the Kremlin, through the media it controls, is conducting a disinformation campaign spreading conspiracy theories about the pandemic and attempting to discredit Western vaccines.[1] For example, the US State Department has accused Moscow in recent weeks of engaging in such actions against US-produced vaccines (by the Pfizer–BioNTech consortium and Moderna). It said that several websites claiming they are harmful had appeared on the Internet at the inspiration of the Russian secret services.[2] Last autumn similar allegations were levelled by the UK authorities: they claimed that Moscow had been engaged in spreading disinformation about the trials for the vaccine developed by AstraZeneca in tandem with Oxford University.[3]
The Kremlin has refuted the Western accusations, and in response blames the US for trying to discourage other countries, including Brazil, from buying Sputnik V, although the Brazilian foreign ministry has denied this report.[4] Furthermore, Moscow has accused all those who raise doubts about the Russian vaccine of being biased against Russia and politicising the issue at the expense of the interests of EU citizens.
Sputnik V: permission to sell
The short-term goal of the Russian campaign is to ensure approval for Sputnik V to be marketed abroad, especially in the European Union countries. In Russia, the vaccine was registered on 11 August and rolled out on 8 September, that is, even before the phase III clinical trials were started in September 2020. On 2 February, preliminary results of these trials were published in the Lancet journal (final data is expected in the coming months). According to these figures, the vaccine’s efficacy was estimated at 91.6%. This publication is basically the only publicly available information on the product. Scientists have drawn attention to several inaccuracies in the study, while also complaining about a lack of access to more detailed data, especially the protocols for the trials.[5]
Outside Russia, Sputnik V was first registered for use by Belarus (21 December 2020). Since then, 60 countries (including Russia itself, as of 12 April 2021) have approved the vaccine. In most cases, this has been done under a fast-track procedure for medicinal products, mainly based on the clinical trials conducted in Russia. Sputnik V has primarily been authorised by post-Soviet states, countries in Latin America, the Arabian peninsula, Asia and Africa. In Europe, it has been registered by Hungary, Slovakia, Serbia, Montenegro and North Macedonia, as well as the Bosnian Serb Republic. In most of them it is mainly competing with Chinese products, as for the time being the Western pharmaceutical companies are focused on fulfilling orders to Western countries. In addition, most of the countries where Sputnik V has been registered are developing countries which are waiting for supplies under the WHO-coordinated COVAX solidarity programme (which Russia has not joined).
In late January 2021, the state-owned Russian Direct Investment Fund (RDIF), responsible for the marketing of Sputnik V, announced it had submitted an application with the EU to authorise the vaccine. However the EMA denied this in early February, stating that no such application had reached it. It turned out that the RDIF had sent it through the portal of the Heads of Medicines Agencies (HMA), which is not involved in the procedure for authorising COVID-19 vaccines.[6] Finally, on 4 March, the EMA started the first phase of the so-called rolling review of Sputnik V, which evaluates data from laboratory studies for efficacy, safety and quality. However, this does not represent the start of the process of registering the product to authorise its marketing. In April, EMA experts are due to inspect the manufacturing facilities in Russia. In February, the agency also began evaluating the German vaccine CureVac and the US vaccine Novavax.
The EMA is calling on EU member states to refrain from registering Sputnik V at the national level until the agency has completed the process of verifying the safety and efficacy of the vaccine and testing it for compliance with European standards (as with the Western preparations). In mid-March, the chair of the EMA’s Management Board Christa Wirthumer-Hoche went as far as to compare the decisions by Budapest and Bratislava to authorise the marketing of Sputnik V to a game of ‘Russian roulette’. In response, Moscow accused her of being biased against the product. The Kremlin spokesman Dmitri Peskov called her statement inappropriate and regrettable, the creators of the Russian vaccine questioned the EMA’s neutrality and demanded an official apology from Wirthumer-Hoche, while RDIF head Kirill Dmitriev accused the agency of deliberately delaying the product’s registration process.
Production of the vaccine
All the countries that have registered Sputnik V are open to its supplies. However, due to Russia’s limited manufacturing capacities, only orders to selected countries are being fulfilled for the time being. From the outset, there has also been no reliable information on the scale of the vaccine’s production. On 5 March, Deputy Prime Minister Tatyana Golikova announced that 13.9 million sets (of two different doses, making up 27.8 million units) had been manufactured, of which 7.9 million (i.e. 15.8 million units) had been rolled out. In February, the output was 7 million sets. In April, however, Aleksandr Ginzburg, head of the Gamaleya Institute (which invented Sputnik V), said that only just over 10 million doses had been rolled out by the end of March.[7] In addition, two more vaccines were launched on the Russian domestic market in March (EpiVacCorona and CoviVac), but so far only around 500,000 doses of the former have been manufactured. Industrial production is not due to start until April.
According to the original assumptions, export shipments Sputnik V were to come from plants outside Russia. Facing serious problems with mass production within the country, the government has been open to transferring the technology to third countries. So far, production has started in Kazakhstan (200,000 doses had been produced by 25 March), and recently at facilities in Belarus (no information on volume is available). Moreover, preparations for mass production are under way in South Korea (the contract is for up to 650 million doses, but the start of production has been postponed), India (850 million doses, as of Q3 2021), Brazil (150 million doses for the Brazilian market in 2021, but due to the lengthy registration procedure the start of production has now been postponed until mid-2021), and also China (the contract is for 60 million doses; production is scheduled to start in May).
In March, Russia succeeded in signing the first agreement to start production on the EU territory, in Italy. According to the Russian side, the contract signed between the RDIF and the Italian pharmaceutical company Adienne, owned by Swiss-based Adienne Pharma & Biotech, stipulates that production will start in July at factories in Lombardy, with 10 million doses to be manufactured by the end of the year. Adienne itself has declined to provide information on the planned production volume, but indicated that the first trial batch could be made in the third quarter of 2021, while production of commercial batches will only start after the Italian Medicines Agency has given its approval. As a result, public pressure has increased on the Italian government, which learned about the deal from the media only after it had been signed. The regional authorities of Campania also announced the signing of a preliminary contract to import the vaccine. The public is demanding that vaccinations be speeded up, and Sputnik V is being touted as a solution to the problem. Russia has also signed a contract with Serbia’s Torlak Institute of Virology and Vaccines: in this case, however, it does not involve the production, but the vialisation of the Russian product. The institute is expected to roll out the first batch (about 4 million doses) in May.
The RDIF has also announced that agreements to produce Sputnik V have been concluded with pharmaceutical companies from Spain, France and Germany, although these reports have been denied by the French and Spanish ministries of commerce. The Italian example shows, however, that governments may not necessarily be informed about cooperation between pharmaceutical companies and the RDIF. Several federal and regional politicians in Germany have made positive comments about the possible production of Sputnik V, including the state secretary at the Ministry for Economic Affairs and Energy Andreas Feicht (CDU), who is responsible for expanding the infrastructure for producing the vaccine in Germany. He mentioned the ongoing talks on the matter which are being held in Illertissen, Bavaria with R-Pharm Germany GmbH (a subsidiary of the Russian biotech company Pharm Group).
Sputnik V exports
Media reports on Sputnik V exports indicate that over 8 million doses have been delivered to its recipients abroad so far. The main source of these reports are the media in the importing countries, as the Russian authorities have not published the relevant statistics.
In the case of Europe, the Kremlin initially targeted the markets of the non-EU countries with which it had good political relations, but which were not covered by joint vaccine purchases, i.e. the Balkan states. Serbia was the first to receive Sputnik V, with a total of over 340,000 out of the 2 million contracted doses delivered by 29 March (this represented around 12% of all the vaccines delivered to Serbia, with the largest shipments arriving from China), although the order is running behind schedule. As a result, the country has one of the highest vaccination rates in Europe, with around 25% of its population having received the first dose by 12 April and 17% having received both (see table below). Smaller amounts have also reached North Macedonia, Montenegro, Albania, San Marino and the Bosnian Serb Republic. When problems arose with implementing the contracts Western pharmaceutical companies had signed with EU member states, Russia began to fight for this market as well. Eventually, Hungary ordered 2 million doses, and received a total of about 1.1 million by 12 April, meaning that Sputnik V accounted for about 23% of all the vaccines delivered to the country; under the contract Budapest should have received 1.6 million doses of the Russian product by March 21. Hungary is the country with the highest vaccination rate in the EU: more than 32% of the population have received one dose, and almost 13% have received two. Slovakia has also decided to purchase Sputnik V (2 million doses, with delivery by June this year). It has so far received 200,000 doses, i.e. about 18% of the total amount of vaccines delivered to the country, but the preparation has not yet been used as it is still undergoing laboratory tests. However, controversies surrounding Prime Minister Igor Matovič’s decision to import the Russian product sparked a government crisis in Slovakia in late March. Moreover, in April the Slovak media published the full version of the national drug control authority (ŠÚKL)’s opinion on the supplied doses of Sputnik V. The agency found that the vaccines received were not identical either with the product being evaluated by the EMA or the one described in the Lancet. According to ŠÚKL, the Russian vaccines used in different countries around the world “have only the name in common”, and the differences involve fundamental issues such as composition or method of storage. ŠÚKL pointed out that despite repeated requests, the Russian side had failed to provide about 80% of the necessary information.
By 12 April, a total of more than 102 million doses of EMA-authorised Western vaccines had reached all the EU member states, while deliveries of Sputnik V to Hungary and Slovakia had totalled around 1.3 million doses. Its largest quantities, meanwhile, have been shipped to Latin America (see table), where the Kremlin has been for years seeking to strengthen its influence, and where local authorities have failed to secure speedy deliveries of the Pfizer–BioNTech vaccine.
Table. Vaccination campaigns in major importers of Sputnik V* (as of 12 April)
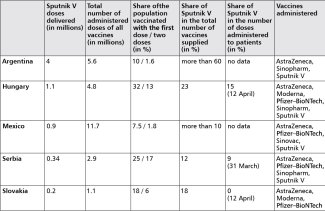
* In Europe, small shipments of Sputnik V have also arrived to the Bosnian Serb Republic (42,000 doses), Albania (10,000), San Marino (7500), Montenegro (7000) and North Macedonia (3000).
Sources: numbers of Sputnik V doses delivered – the author’s own calculations; COVID-19 Vaccine Tracker, European Centre for Disease Prevention and Control, www.ecdc.europa.eu; COVID-19 Tracker – Hungary, Reuters Graphics, graphics.reuters.com.
The Russian vaccine divides the EU
In the European Union, a variety of opinions can be heard on the wisdom of using the Russian jab. For example, numerous calls to approve its rollout in the EU as soon as possible and start deliveries & production have come from Germany. The case made by the authorities of some German federal states focuses on the alleged availability of Sputnik V in the face of problems experienced by other suppliers, its higher efficacy than some of the vaccines used in the EU, and the lower incidence of adverse vaccine reactions. A number of German politicians have been demanding that the European Commission start negotiations on the purchase of the Russian product before the EMA approves its use. Similar statements have also been made by individual Italian regions which have expressed a desire to import Sputnik V. Greece, in turn, without waiting for the outcome of the internal EU debate on Covid passports, has announced that it will recognise the vaccinations of Russian tourists, which would de facto represent Athens’ partial recognition of the vaccine.
On the other hand, Karl Lauterbach (the SPD’s health spokesman) has expressed scepticism about the reporting of possible post-vaccination effects, pointing out that the Gamaleya Institute’s claims of an insignificant number of such cases are ‘unrealistic’. For his part, the EU’s internal market commissioner Thierry Breton has stated that Brussels does not need Sputnik V and will prioritise the four Western jabs already authorised, the quantities of which should ensure herd immunity in the EU by this July. He has also highlighted Russia’s problems with producing the vaccine, and has left open the possibility that the EU could make the capacity of one of its factories available for this purpose at a later stage. Lithuania’s Prime Minister Ingrida Šimonytė has even called Sputnik V ‘Putin’s hybrid weapon’. French President Emmanuel Macron has accused Russia and China of using vaccines to strengthen their influence and described their policy in this area as a de facto ‘new world war’. In this context, he has emphasised the need to strengthen the EU’s independence and develop its own production capacities. Meanwhile, the head of the European Commission Ursula von der Leyen has expressed her bewilderment at Russia’s willingness to implement foreign contracts worth millions for the supply of Sputnik V at a time when the vaccination campaign in Russia itself is proceeding slowly. The percentage of people vaccinated with the first dose in Russia is only around 6.1%, compared to 16.8% in the EU countries taken together, 36% in the US, and over 48% in the UK.
Moscow is trying to fend off the allegations. RDIF head Kirill Dmitriev has said that claims that Russia is politicising the process of exporting Sputnik V are being made by the country’s opponents who consistently express this dislike in public, which prevents them from taking an objective view of reality, i.e. recognising that Russia is one of few countries that have succeeded in developing an effective vaccine for which demand significantly exceeds supply.[8] In mid-March, Western media reported that despite its officially expressed distance from Sputnik V, the European Union was now in behind-the-scenes talks with Moscow about it.[9] However, the European Commission has denied these rumours.
Summary and conclusions
The difficult epidemiological situation in Europe – combined with a relatively slow vaccination campaign in the EU compared with Israel, the United Kingdom or the US – is generating social discontent, and is consequently raising the pressure on European politicians to urgently find more effective ways of tackling the pandemic. As a result, we have seen blame-shifting in the EU for the existing situation and efforts to find all kinds of solutions, such as importing vaccines which are yet to be authorised. The Kremlin is trying to take advantage of this to step up its promotion of Sputnik V and urge EU member states to buy it. In doing so, the Russian side is creating the impression that its preparation could solve the EU’s current problems with vaccine shortages, while in fact any major deliveries would be highly unlikely to begin until the second half of 2021, when – according to EU forecasts – they will no longer be needed as the Western products will meet the demand. Moreover, deliveries of the quantities of Sputnik V already contracted (including to Serbia, Hungary and Argentina) are running behind schedule. Ambitious production plans, especially for territories outside the Russian Federation (India, South Korea, Brazil), are also being pushed back. Russia has only managed to export a total of more than 5 million doses of Sputnik V so far, while a total of more than 80 million vaccines from Western manufacturers have already reached EU member states (as of 29 March).
The Kremlin is de facto using the Russian-made vaccine as a soft power instrument to improve its image in the European Union and to disrupt its unity, particularly between the member states and Brussels. Russian accusations that the EMA is biased may pose a major challenge to EU cohesion, especially in the context of strong pressure from several EU states to complete the Sputnik V verification process as soon as possible and allow it to be imported. It remains to be seen whether under these circumstances the agency will be able to adhere to all its control procedures if, for example, it decides that Phase III clinical trials should be repeated in the EU (thus delaying its certification by months), and whether more member states will choose to ignore its recommendation and register the vaccine at the national level after all.
It should be stressed that Russia does not currently have enough stocks of Sputnik V to realistically speed up its vaccination campaign in Europe. The situation can only change with the expected start of production in South Korea (650 million doses in 2021–2022): the first shipment had been scheduled for March but did not take place. In February, it was reported that a trial batch was due to arrive in Moscow, but so far there is no word as to whether it has been accepted and mass production can begin. Significant amounts of Sputnik V are unlikely to be rolled out before this autumn.
The text was updated on 13 April 2021.
[1] For more, see ‘Attacking the West, putting Russians in danger’, EUvsDisinfo, 29 March 2021, www.euvsdisinfo.eu.
[2] M.R. Gordon, D. Volz, ‘Russian Disinformation Campaign Aims to Undermine Confidence in Pfizer, Other Covid-19 Vaccines, U.S. Officials Say’, The Wall Street Journal, 7 March 2021, www.wsj.com.
[3] ‘Reported Russian COVID vaccine disinformation 'reprehensible' – Raab’, Reuters, 16 October 2020, www.reuters.com.
[4] A.N. Farzan, H. Traiano, ‘U.S. officials pushed Brazil to reject Russia’s coronavirus vaccine, according to HHS report’, The Washington Post, 16 March 2021, www.washingtonpost.com.
[5] E. Bucci, ‘More concerns on the “Sputnik” vaccine’, Cattivi Scienziati, 9 February 2021, www.cattiviscienziati.com.
[6] It was a surprising mistake, especially as the Gamaleya Institute had previously said that experts from Germany’s Paul Ehrlich Institute had been consulted on the EU registration procedure. A. Zverev, P. Ivanova, ‘Confusion over Russia's EU vaccine approval bid could be result of misdirected application’, Reuters, 19 February 2021, www.reuters.com.
[7] Ginzburg did not specify whether he meant a dose as a set of two units. In Sputnik V export contracts, the term is usually used, as with Western vaccines, to describe a unit of vaccine needed for a single administration to a patient.
[8] Г. Полонская, ‘Кирилл Дмитриев: «Вакцины должны быть вне политики»’, EuroNews, 5 March 2021, ru.euronews.com.
[9] F. Guarascio, J. Chalmers, E. Parodi, ‘Unthinkable? EU considers getting a vaccine boost from Russia's Sputnik’, Reuters, 15 March 2021, www.reuters.com.





